The Role of Complement Activation in Driving Myeloid-mediated Immunosuppression in Pancreatic Cancer
We are studying the role of the complement cascade, an ancient and highly conserved part of the innate immune system, in pancreatic cancer. We have found that complement activation promotes myeloid-mediated immunosuppression in the tumor microenvironment. Our ongoing work focuses on determining whether complement inhibition is a viable therapy target for pancreatic cancer.
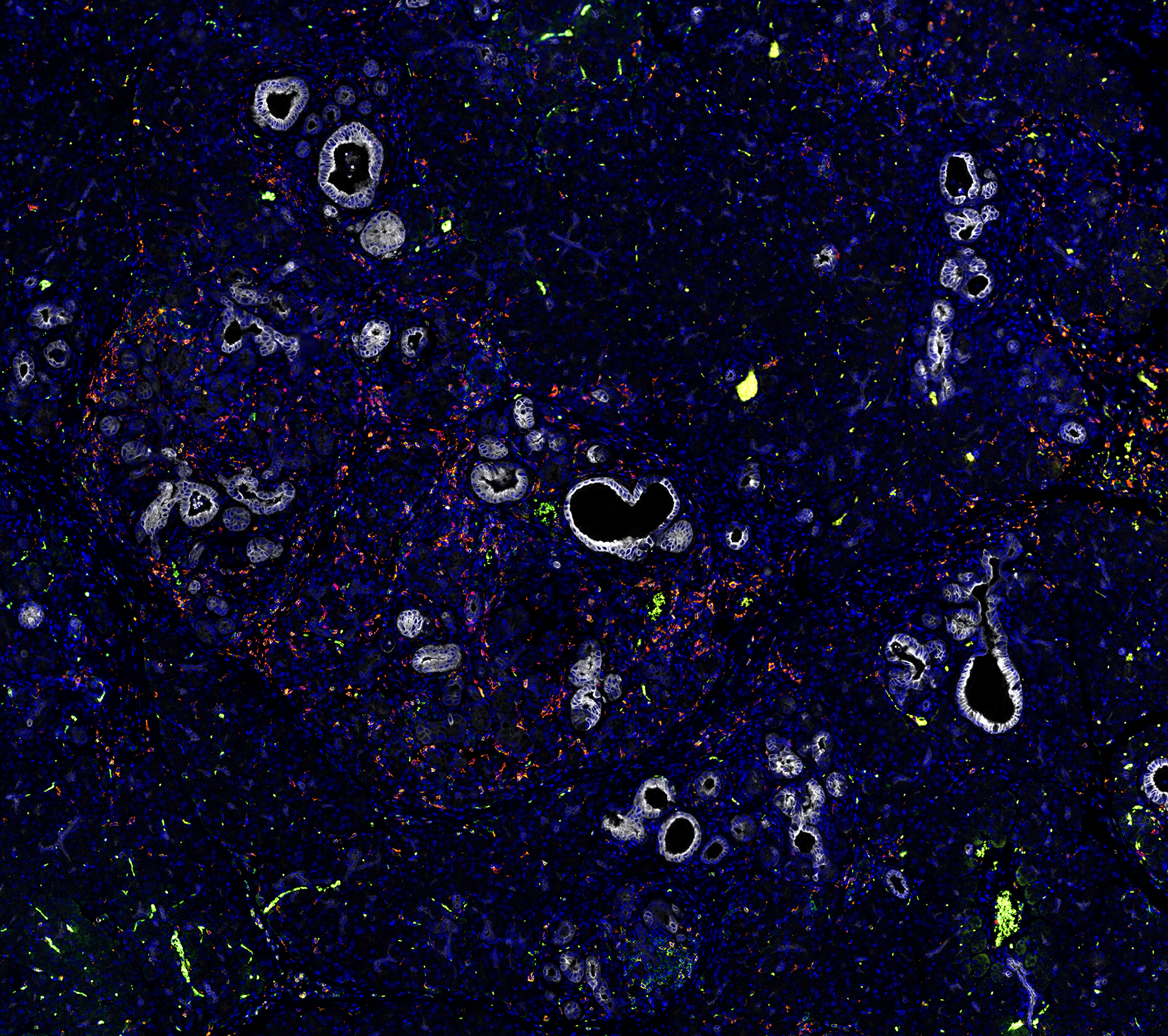
Mouse models of pancreatic cancer provide a unique opportunity to study which non-malignant cells are present in the earliest stages of pancreatic cancer. Pictured above are pre-invasive pancreatic cancer lesions stained to highlight cancer cells (white), T cells (orange/yellow), Macrophages (light blue), and Neutrophils (Red). Image was generated by Alex Jefferson and Emma Dawson.
Investigating the Antigen Landscape and Immune Responses in Mouse PDAC Models
We are studying antigen presentation in pancreatic cancer cells with a particular focus on determining the effects of Kras inhibition on shaping the antigen repertoire and immune response with the goal of designing effective, personalized antigen therapy.
Investigating the Role of T cells in Epigenetic Remodeling during Pancreatitis
There is a growing appreciation that the combination of genetic alterations and external factors, such as inflammation, drives cancer. Pancreatitis, acute or chronic inflammation of the pancreas, is a significant risk factor for developing pancreatic ductal adenocarcinoma (PDAC), one of the deadliest malignancies. Despite research indicating CD4+ and CD8+ T cells infiltrate and modulate inflammatory and fibrotic responses in the pancreas during pancreatitis, the role of T cells in the context of inflammation-induced PDAC initiation and progression is unexplored. Our preliminary findings indicate T cells modulate a key and unique inflammatory-reparative response engaged during pancreatitis, acinar-to-ductal metaplasia (ADM). ADM relies on cellular interactions but is fundamentally an epigenetic process thus we will be investigating the role of T cells in epigenetic remodeling during pancreatitis.
Epigenetic Regulation of Pancreatic Cancer Progression
This project investigates the role of chromatin remodeling in shaping tumor evolution, metastasis, and therapeutic resistance in pancreatic ductal adenocarcinoma (PDAC). Focusing on ARID1A—one of the most frequently mutated chromatin regulators in PDAC—the work combines genetically engineered organoid models with orthotopic transplantation to investigate how the loss of ARID1A reprograms the cell state and promotes disease progression. Ongoing efforts leverage single-cell multiomic approaches to characterize the transcriptional and epigenetic trajectories associated with ARID1A loss. In parallel, an in vivo CRISPR screen targeting chromatin regulators is being developed to identify modulators of metastatic competence. These studies aim to uncover the epigenetic mechanisms underlying phenotypic plasticity and identify context-specific vulnerabilities in aggressive PDAC subtypes.